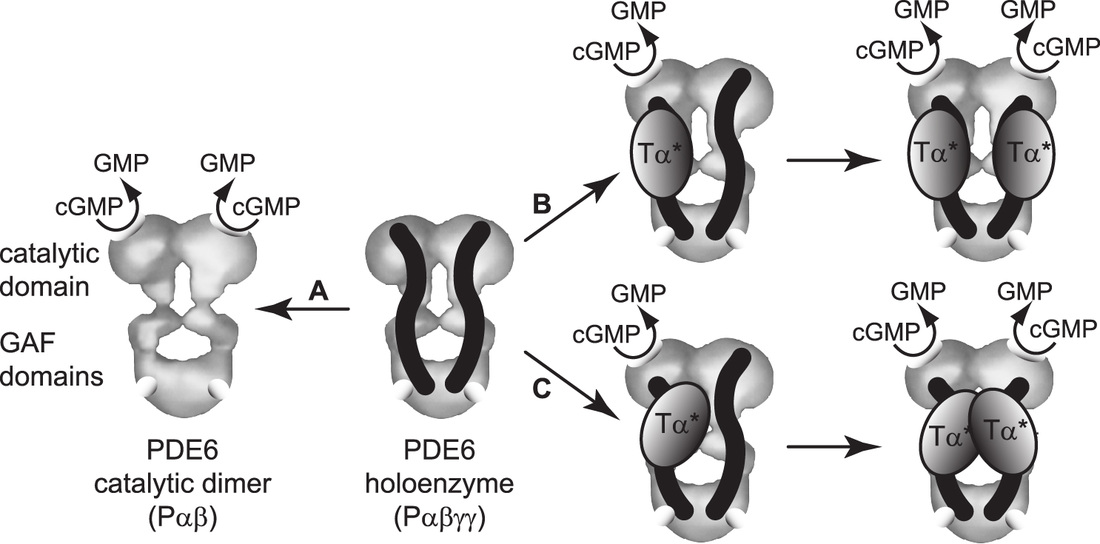
|
|
Many of the following publications are available for full-text downloads on ResearchGate
X. H. Zeng-Elmore, X. Z. Gao, R. Pellarin, D. Schneidman-Duhovny, X. J. Zhang, K. A. Kozacka, Y. Tang, A. Sali, R. J. Chalkley, R. H. Cote, and F. Chu. (2014) Molecular architecture of photoreceptor phosphodiesterase elucidated by chemical cross-linking and integrative modeling. J.Mol.Biol. in press. Karyn B.Cahill, Jonathan H. Quade, Karen L. Carleton, Rick H. Cote. (2012) Identification of Amino Acid Residues Responsible for the Selectivity of Tadalafil Binding to Two Closely Related Phosphodiesterases, PDE5 and PDE6. J.Biol.Chem. 287, 41406-41416 Zhang, X.J., Gao, X.Z., Yao, W., and Cote, R.H. (2012) Functional mapping of interacting regions of the photoreceptor phosphodiesterase (PDE6) γ-subunit with PDE6 catalytic dimer, transducin, and Regulator of G-protein Signaling9-1 (RGS9-1). J.Biol.Chem. 287, 26312-26320 Matte, S.L., Laue, T.M., and Cote, R.H. (2012) Characterization of conformational changes and protein-protein interactions of rod photoreceptor phosphodiesterase (PDE6). J.Biol.Chem. 287, 20111-20121 Cahill, K. B. and Cote, R. H. (2011) Phosphodiesterase 6C, cGMP-specific cone a'. UCSD-Nature Molecule Pages doi:10.1038/mp.a001756.01 Gitschier, H. J. and Cote, R. H. (2011) Phosphodiesterase 6D, cGMP-specific rod d. UCSD-Nature Molecule Pages doi:10.1038/mp.a001757.01 Zhang, X. J. and Cote, R. H. (2011) Pde6H, cone-specific inhibitory subunit of cone PDE6. UCSD-Nature Molecule Pages doi:10.1038/mp.a001758.01 X. J. Zhang, N. P. Skiba, and R. H. Cote. (2010).Structural requirements of the photoreceptor phosphodiesterase γ-subunit for inhibition of rod PDE6 holoenzyme and for its activation by transducin. J.Biol.Chem. 285, 4455-4463 Liu,Y., Matte,S., Corbin,J., Francis,S., and Cote,R.H. (2009). Probing the catalytic sites and activation mechanism of photoreceptor phosphodiesterase (PDE6) using radiolabeled PDE inhibitors. J Biol. Chem. 284, 31541-31547 Cote,R.H. and Cahill,K.B. (2009). Function and regulation of photoreceptor phosphodiesterase (PDE6) in the visual signaling pathway. In Handbook of Cell Signaling, R.Bradshaw and E.Dennis, eds. (San Diego: Elsevier), pp. 1445-1451 Zhang, X.J., Cahill, K.B., Elfenbein, A., Arshavsky, V.Y., Cote, R.H. (2008). Direct allosteric regulation between the GAF domain and catalytic domain of photoreceptor phosphodiesterase PDE6. J Biol. Chem. 283, 29699-29705 Cote ,R.H. (2008). The cGMP signaling pathway in retinal photoreceptors and the central role of photoreceptor phosphodiesterase (PDE6). In Visual Transduction and Non-Visual Light Perception, J.Tombran-Tink and C.J.Barnstable, eds. (Totawa: Humana Press), pp. 141-169. Cote ,R.H. (2006). Photoreceptor phosphodiesterase (PDE6): a G-protein-activated PDE regulating visual excitation in rod and cone photoreceptor cells. In Cyclic Nucleotide Phosphodiesterases in Health and Disease, J.A.Beavo, S.H.Francis, and M.D.Houslay, eds. ( Boca Raton , FL : CRC Press), pp. 165-193. Pentia,D., Hosier,S., and Cote,R.H. (2006). The glutamic acid-rich protein-2 (GARP2) is a high affinity rod photoreceptor phosphodiesterase (PDE6)-binding protein that modulates its catalytic properties. J Biol. Chem. 281: 5500-5505 Zhang,X., Feng,Q., and Cote,R.H. (2005). Efficacy and selectivity of phosphodiesterase-targeted drugs in inhibiting photoreceptor phosphodiesterase (PDE6) in retinal photoreceptors. Invest. Ophthalmol. Vis. Sci. 46 , 3060-3066. Zhang,H., Hosier,S., Terew,J.M., Zhang,K., Cote ,R.H., and Baehr,W. (2005). Assay and functional properties of PrBP (PDEd), a prenyl binding protein interacting with multiple partners. Methods Enzymol. 403: 42-56 Cote,R.H. (2005). Cyclic guanosine 5'-monophosphate binding to the regulatory GAF domains of photoreceptor phosphodiesterase (PDE6). Methods in Molecular Biology 307 , 141-154. Pentia,D.C., Hosier,S., Collupy,R.A., Valeriani,B.A., and Cote,R.H. (2005). Purification of PDE6 isozymes from mammalian retina. Methods in Molecular Biology 307: 125-140. Carleton, K.L., Spady, T.C., and Cote, R.H. (2005). Rod and cone opsin families differ in spectral tuning domains but not signal transducing domains as judged by saturated evolutionary trace analysis. J. Mol. Evol. 61: 75-89 Zhang, X. and Cote, R.H. (2005) cGMP signaling in vertebrate retinal photoreceptor cells. Front. Bioscience 10:1191-1204 Norton AW, Hosier S, Terew JM, Li N, Dhingra A, Vardi N, Baehr W, Cote RH. (2005) Evaluation of the 17 kDa prenyl binding protein as a regulatory protein for phototransduction in retinal photoreceptors. J Biol Chem. 280: 1248-1256 Cote , R.H. (2004) Characteristics of photoreceptor PDE (PDE6): similarities and differences to PDE5. Intl. J. Impotence Res. 16, S28-S33 Corbin, J. D., Kotera, J., Gopal, V. K., Cote, R. H., and Francis, S. H. (2003). Regulation of cyclic nucleotide levels by sequestration. In Handbook of Cell Signaling, R. Bradshaw and E. Dennis, eds. (San Diego: Academic Press), pp. 453-457. Cote, R. H. (2003). Structure, function, and regulation of photoreceptor phosphodiesterase (PDE6). In Handbook of Cell Signaling, R. A. Bradshaw and E. A. Dennis, eds. (San Diego: Academic Press), pp. 453-458. Paglia, M. J., Mou, H., and Cote, R. H. (2002). Regulation of photoreceptor phosphodiesterase (PDE6) by phosphorylation of its inhibitory γ subunit re-evaluated. J. Biol. Chem.277, 5017-5023. Kameni Tcheudji, J. F., Lebeau, L., Virmaux, N., Maftei, C. G., Cote, R. H., Lugnier, C., and Schultz, P. (2001). Molecular organization of bovine rod cGMP-phosphodiesterase 6. J. Mol. Biol. 310, 781-791. Mou, H. and Cote, R. H. (2001). The catalytic and GAF domains of the rod cGMP phosphodiesterase (PDE6) heterodimer are regulated by distinct regions of its inhibitory γ subunit. J. Biol. Chem. 276, 27527-27534. Cote, R. H. (2000). Kinetics and regulation of cGMP binding to noncatalytic binding sites on photoreceptor phosphodiesterase. Methods Enzymol. 315, 646-672. Norton, A. W., D'Amours, M. R., Grazio, H. J., Hebert, T. L., and Cote, R. H. (2000). Mechanism of transducin activation of frog rod photoreceptor phosphodiesterase: allosteric interactions between the inhibitory γ subunit and the noncatalytic cGMP binding sites. J. Biol. Chem. 275, 38611-38619. D'Amours, M. R. and Cote, R. H. (1999). Regulation of photoreceptor phosphodiesterase catalysis by its noncatalytic cGMP binding sites. Biochem. J. 340, 863-869. D'Amours, M. R., Granovsky, A. E., Artemyev, N. O., and Cote, R. H. (1999). The potency and mechanism of action of E4021, a PDE5-selective inhibitor, on the photoreceptor phosphodiesterase depends on its state of activation. Mol. Pharmacol. 55, 508-514. Mou, H., Grazio, H. J., Cook, T. A., Beavo, J. A., and Cote, R. H. (1999). cGMP binding to noncatalytic sites on mammalian rod photoreceptor phosphodiesterase is regulated by binding of its γ and δ subunits. J. Biol. Chem. 274, 18813-18820. Artemyev, N. O., Arshavsky, V. Y., and Cote, R. H. (1998). Photoreceptor phosphodiesterase: Interaction of inhibitory γ subunit and cyclic GMP with specific binding sites on catalytic subunits. Methods 14, 93-104. Hebert, M. C., Schwede, F., Jastorff, B., and Cote, R. H. (1998). Structural features of the noncatalytic cGMP binding sites of frog photoreceptor phosphodiesterase using cGMP analogs. J. Biol. Chem. 273, 5557-5565. Coccia, V. J. and Cote, R. H. (1994). Regulation of intracellular cyclic GMP concentration by light and calcium in electropermeabilized rod photoreceptors. J. Gen. Physiol. 103, 67-86. Cote, R. H., Bownds, M.D., and Arshavsky, V. Y. (1994). cGMP binding sites on photoreceptor phosphodiesterase: Role in feedback regulation of visual transduction. Proc. Natl. Acad. Sci. U.S.A. 91, 4845-4849. Cote, R. H. and Brunnock, M. A. (1993). Intracellular cGMP concentration in rod photoreceptors is regulated by binding to high and moderate affinity cGMP binding sites. J. Biol. Chem. 268, 17190-17198. Forget, R. S., Martin, J. E., and Cote, R. H. (1993). A centrifugal separation procedure detects moderate affinity cGMP binding sites in membrane-associated proteins and permeabilized cells. Anal. Biochem. 215, 159-161. Lisman, J. E., Erickson, M., Richard, E. A., Cote, R. H., Bacigalupo, J., Johnson, E. C., and Kirkwood, A. (1992). Mechanisms of amplification, deactivation and noise reduction in invertebrate photoreceptors. In Sensory Transduction, D. P. Corey and S. D. Roper, eds. (New York: Rockefeller Press), pp. 176-199. Cote, R.H., Nicol, G.D., Burke, S.A., and Bownds, M.D. (1989). Cyclic GMP levels and membrane current during onset, recovery and light adaptation of the photoresponse of detached frog photoreceptors. J. Biol. Chem. 264, 15384-15391 Robinson, P.R. and Cote, R.H. (1989). Characterization of guanylate cyclase in squid photoreceptors. Vis. Neurosci. 3, 1-7 Bownds, M.D., Nicol, G.D., and Cote, R.H. (1987). Biochemical pathways mediating excitation and adaptation of vertebrate rod photoreceptor cells. In Retinal Proteins, Y.A.Ovchinnikov, ed. VNU Science Press, pp. 95-108 Cote, R.H., Nicol, G.D., Burke, S.A., and Bownds, M.D. (1986). Changes in cGMP concentration correlate with some, but not all, aspects of the light-regulated conductance of frog rod photoreceptors. J.Biol. Chem. 261, 12965-12975 Robinson, P.R., Radeke, M.J., Cote, R.H., and Bownds, M.D. (1986). cGMP influences guanine nucleotide binding to frog photoreceptor G-protein. J. Biol. Chem. 261, 313-318 Cote, R.H., Biernbaum, M.S., Nicol, G.D., and Bownds, M.D. (1984). Light-induced decreases in cGMP concentration precede changes in membrane permeability in frog rod photoreceptors. J. Biol. Chem. 259, 9635-9641 Cote, R.H., Anderson, C.F., and Borisy, G.G. (1981). Formulation of the general rate equation for subunit flux at steady-state. J. Mol. Biol. 150, 599-602 Cote, R.H., and Borisy, G.G. (1981). Head-to-tail polymerization of microtubules in Vitro. J. Mol. Biol. 150, 577-602 Abe, M., Brown, C., Hendrickson, W.G., Boyd, D.H., Clifford, P., and Cote R.H. (1977). Release of Escherichia coli DNA from membrane complexes by single-strand endonucleases. Proc. Natl. Acad. Sci. U.S.A. 74, 2756-2760. Sonenshein, A.L., Cami, B., Brevet, J., and Cote, R.H. (1974). Isolation and characterization of rifampin-resistant and streptolydigin-resistant mutants of Bacillus subtilis with altered sporulation properties. J. Bacteriol. 120, 253-265 |