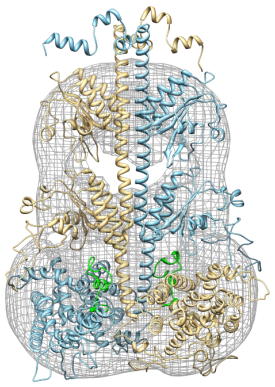
PDE6 is a tetrameric protein The rod PDE6 holoenzyme is a tetramer consisting of homologous α and β catalytic subunits which form a dimer and to which two inhibitory γ subunits bind. Cone PDE6 differs from rod PDE6 in having a catalytic dimer of two identical α subunits to which two cone-specific inhibitory β subunits bind. The functional consequence of rod and cone subunits having somewhat different amino acid sequences is currently not well understood.
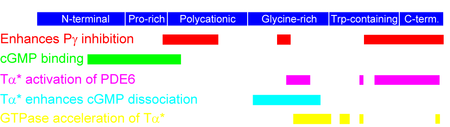
Electron microscopic analysis of purified rod PDE6 catalytic dimers at 2.8 nm resolution reveals a 3-D structure comprised of three distinct globular domains (Kameni Tcheudji et al., 2001). The largest is the catalytic domain, while the two smaller domains correspond to the tandem GAFa-GAFb domains. The primary site of dimerization is between the GAFa domains of two catalytic subunits. PDE6 regulated by inhibitory γ-subunits Each inhibitory γ-subunit interacts with multiple regions of the catalytic subunits, and the affinity of γ binding is regulated by cGMP binding to the GAF domains of the catalytic subunit (Norton et al., 2000; Mou and Cote, 2001). The C-terminal residues of the 87 amino acid γ subunit bind directly to the active site within the catalytic domain (Granovsky et al., 1997). The N-terminal half of γ binds to the catalytic dimer with 50-fold higher affinity than its C-terminal half, and is responsible for the cGMP-dependent modulation of γ affinity by the GAF domain (Mou and Cote, 2001). The C-terminal half of γ also allosterically communicates with the catalytic dimer upon interaction of PDE6 with activated transducin (Zhang et al., 2012
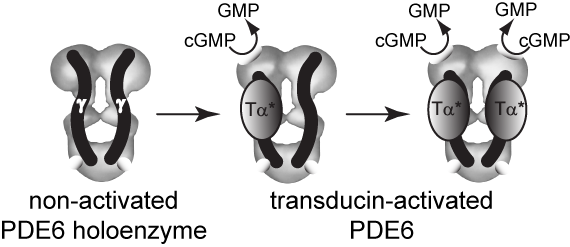
Transducin activates PDE6 by binding the γ-subunit and relieving inhibition
Upon light activation of rhodopsin during visual excitation, activated transducin (αt*-GTP) binds to the PDE6 holoenzyme. PDE6 activation results from physical displacement of its inhibitory γ subunit from the active site. Because transducin can activate rod PDE6 to only 50% of the activity of purified catalytic dimers, it is likely that only one of the two catalytic sites is subject to activation by transducin during phototransduction. Furthermore, only under conditions where cGMP dissociates from the GAF domains of PDE6 will the γ subunit completely dissociate from the PDE6 catalytic dimer (D'Amours and Cote, 1999; Norton et al., 2000).
|
|